Iron charge of see9/12/2023 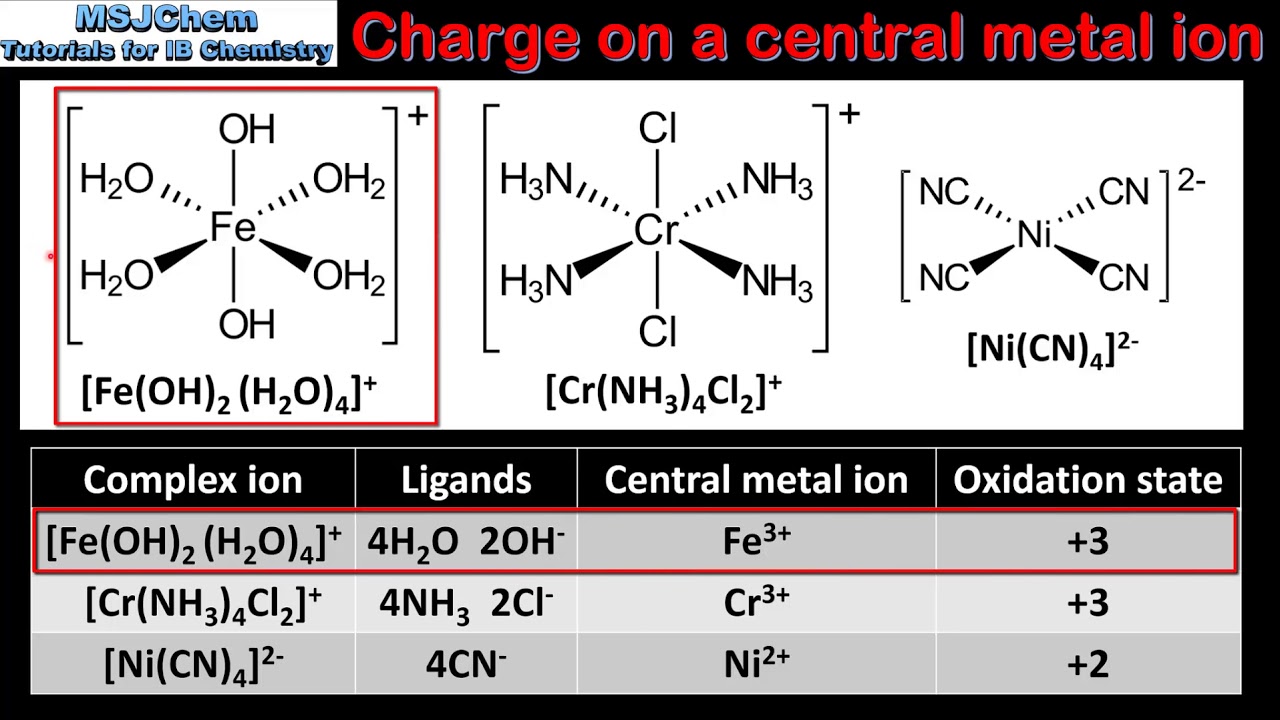
The name of the ionic compound formed from magnesium and nitrate ions is magnesium nitrate and the formula is Mg(NO3)2 M g ( N O 3 ) 2. What can form an ionic compound with magnesium? Sulfur, which only needs two electrons to complete it octet, will pick up the two electrons coming from magnesium, becoming the sulfide anion, S2−, in the. The name of the ionic compound formed from magnesium and nitrate ions is magnesium nitrate and the formula is Mg(NO3)2 M g ( N O 3 ) 2.256.4065 :: Chemistry Applications::Chemistry Applications:: » Chemical Elements, Periodic Table » Compound Name Formula Search 6.8 million yen to usd A: Standard nomenclature rule, also known as IUPAC naming system, is a best method to name any…. Q: Write the names of the following compounds, using the standard nomenclature rule. Acids are compounds containing an ionizable proton (H +), since an acid is a proton donor (a hydrogen atom which has lost its electron).A: Iron (lll) oxide:- Iron (III) oxide or ferric oxide is the inorganic compound. Prefixes are only used for covalent compounds formed from non-metal elements. Molar Mass: 148.3148Please note that ionic compounds (Type I & II binary compound names) never use prefixes to specify how many times an element is present. To name a monatomic anion change the suffix of the element's name to.Magnesium Nitrate Mg(NO3)2 Molar Mass, Molecular Weight. Magnesium dinitrate is a hygroscopic, crystalline solid white.What is the compound name of MgNO32? Wiki User. Magnesium nitrate is also called Magnesium dinitrate or Magniosan or Nitromagnesite (hexahydrate). 25malx ' Mg (NO 3) 2 is an inorganic nitrate salt of magnesium with a chemical name Magnesium nitrate. The elements are magnesium (Mg), nitrogen (N) and oxygen (O). Since you already have the chemical formula of the compound, it is not difficult to make up what elements it is made up from.materials: rubberised cork composition sheet, is 4253, 1980, part ii, grade: rc 70c. 25623299supply of, gasket for 100mm tfp valve of thickness: 06mm as per cee drg. You do not use the prefix di for the nitrate since this is an ionic compound, and it can only be (NO3)2 as nitrate has a -1 charge and Mg. Here some of you may think that iron may also show $ - 4$ oxidation to fulfill the d-orbital and attain highest stability, but this assumption is not correct because energy required to gain four electrons is high as compared to removing two or three electrons.Mgno32 compound name 256.4065 :: Chemistry Applications::Chemistry Applications:: » Chemical Elements, Periodic Table » Compound Name Formula Search As we know that atomic number of iron is 26 & its electronic configuration is written as $\left[ $ orbital of iron atom to get more stability because in the half filled condition d-orbital shows great stability, and due to this iron will show $ + 3$ oxidation state. Iron have two oxidations state because of the below reason: And negative oxidation number which shows by the atom when they accept electrons on the outermost shell from the surrounding. Positive oxidation number which shows by the atom when they lose electrons from the outermost shell. Oxidation number are present in two types and they are: Hint: First of all we have to know about the oxidation state and oxidation state of any atom will define as the number of electrons gained or lost by that atom with another species from its outermost shell.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
